
Glenorchy
Tasmania 7010 Australia
Phone: 61 3 62497868 (am)
Phone: 61 3 62713000 (pm)
Fax: 61 3 62730010
www.tececo.com
 |
497 Main Road Glenorchy Tasmania 7010 Australia Phone: 61 3 62497868 (am) Phone: 61 3 62713000 (pm) Fax: 61 3 62730010 www.tececo.com |
![]() Printed in cyberspace on recycled electrons
Printed in cyberspace on recycled electrons
Keeping you informed about TecEco sustainability projects.ĀIssue 84, 5 January 2008
There has been another rash of publicity from a British company called Novacem which is a spin off from Imperial College London claiming they have discovered some new additives that can be added to reactive magnesia causing it to more rapidly carbonate. We don't mind people trying to top us technically if their invention is genuine, but to claim the concept of carbonation in magnesium binders as a means of curtailing global warming as their idea in their publicity campaign is totally dishonest, especially as John Harrison introduced them to it. It angers us that Cambridge/Imperial College working together have chosen to try and steal our thunder on this rather than try an work with us as the need for solutions is urgent and must be found with the next few years. The following article discusses some of the wacky solutions that are being proferred and there is little doubt that the concept of using man made carbonate is clearly the most viable.
At this point in time we do not know whether the invention is genuine or whether it is an existing idea for carbonation rehashed for concrete. We we strongly suspect the latter. A new application for an existing technology is not patentable and there are a host of substances that could be added to accelerate or catalyse carbonation that are prior art and therefore not patentable. Furthermore whatever the additive Novacem are using is, it will end up buried in concrete so it has to be cheap. We chose Portland cement as our main catalyst/accelerator because it is cheap and universally available and when buried in concrete it adds considerable strength. As carbonation even with relatively small proportions added in permeable substrates ensues rapidly we cannot understand the determined effort by the Cambridge University - Imperial College London researchers to first discredit us and then try and steal our concept and reinvent our technology. We would like to think otherwise but have no choice but to conclude that their behaviour is very unethical. More details are to be found at http://localhost/TecEco/rdandd.thirdparty.php
This page was first written as an article drawing heavily on a 1996 paper by Dr Ben Matthews[1] and the original is still downloadable from our web site here. It has been updated as a web page mainly because of the obvious failure of the Kyoto process and the large number of articles starting to appear in credible newspapers and from leading institutions and people calling for radical action to prevent what is shaping up to be runaway global warming by 2020 or when the ice all melts - whichever comes first.
Many of the articles we have read giving alternative technical solutions to global warming do not as Ben now puts it on his web site (chooseclimate.org) sufficiently distinguish between "realistic" and "wild" or wacky ideas. Dr Matthews accepts the need for an update and has been collecting ideas. We look forward to this however in the meantime we are publishing our expanded version here. Because there was some need to focus many of the more wacky ideas in Ben's paper or that have emerged since are not included as just too wacky and some new ones have been included.
As far back as a year ago there was public recognition of the importance of technological fixes and the World Business Council for Sustainable Development and the International Chamber of Commerce issued a Communique from Bali that at point 4 said "Technology is key, for addressing the climate challenges. There is a need for scaling up of R&D jointly between Governments and Business as well as accelerating the deployment of technologies" (See Newsletter75)
More recently an article appeared in The Independent of Friday 2 January 2009 [2] in which their science editor Steve Conner assisted by a Chris Green make it quite clear that "An emergency "Plan B" using the latest technology is needed to save the world from dangerous climate change". The Independent undertook a poll of "80 international specialists in climate science" and determined that "what has worried many of the experts, who include recognised authorities from the world's leading universities and research institutes, as well as a Nobel Laureate, is the failure to curb global greenhouse gas emissions through international agreements, namely the Kyoto Treaty, and recent studies indicating that the Earth's natural carbon "sinks" are becoming less efficient at absorbing man-made CO2 from the atmosphere."
We agree with The Independent authors that the Kyoto process or any like it has not and will not work because constraint or substitution have perceived and real costs and besides, it would seem obvious that although desirable, rapidly decoupling the economy from fossil fuels would cause economic mayhem. (Please read our economics page and political analysis ).
The greatest challenge civilisation has ever faced is gathering momentum and given the incredible dangers of adopting the wrong technology platforms to address it we advocate that all proposed solutions pass two simple basic tests:
We think the solution should be to change the way we do things in ways that promote rather than limit economic activity and that of course meet the above criteria. Below is a summary of all the different ideas out there in table form and the following text fills out the detail.
We conclude that there are only too clear winners. Improving the management of our soils so soil carbon is increased and learning to make and use man made carbonates as first proposed by John Harrison in 2002 .
Basic Strategy |
Technical Solution |
Sub Category of Technical Solution |
Advantages and Practicality |
Problems/Risks |
Cost/ Profit? |
Our Rating(1 totally wacky, 10 excellent chance of success) |
Increasing energy efficiency |
Obvious advantages of doing more with less. | With heat engines (e.g. car engines) further opportunities for improving efficiency are limited. | Increases in efficiency save energy and are thus profitable depending on the cost of achieving the improved level of efficiency. Law of diminishing returns starting to apply. See Di Fazio [16] ) | 4 | ||
Using renewable energy |
Obvious advantages provided supply and cost issues can be overcome | Many forms of alternative energy do not have the energy density of gasoline or do not work 24/7 to produce base load power. | Many alternative energy solutions are not competitive with fossil fuels particularly for base load and transport energy. The rate of technical improvement and uptake are both greater than that for heat engines such as power station turbines and will increase with a legal price on carbon emissions resulting in greater economies of scale further accelerating uptake.. | 7 | ||
Modifying the CO2 balance as a result of geo or planetary engineering |
General principles | We have in fact been unknowingly affecting climate for thousands of years and now have no choice but to modify our activities. | Runaway population growth coupled with dependency on fossil fuels for energy. | TecEco believe it is possible to change the physical basis of the economy profitably. | ||
Climate engineering proposals involving the reflection of incoming solar radiation |
General principles | The objective is to reduce the total energy reaching the earth however there are disadvantages associated with the methods compared to Gaia Engineering. | These ideas are essentially wacky science. | Not likely to be achieved on a large enough scale cost effectively to make a difference. | ||
| Injecting the air with particles to reflect sunlight (using aerosols) | Using aerosols would reduce the total energy reaching the earth but the cost and downside risks result in low overall benefits. | May aerosols such as sulfur dioxide used in the quantities required have several other downsides such as in the case of sulfur dioxide - acid rain. | Not likely given the quantities required. | 2 | ||
| Giant reflectors or sun shields orbiting the earth | Not practical at all | The cost of actually putting in the sky enough reflectors and maintaining them would be prohibitive. Besides they would all eventually fall to the earth. | The cost would be astronomical | 1 | ||
| Creating low salt water clouds over the oceans | Not practical on a cost and risk basis | Salt water clouds raining over land would be unacceptable | High cost and inability to control the wind. | 2 | ||
Climate engineering proposals involving removal of CO2 from the air by enhancing natural sinks |
General principles | Increasing the efficiency or capacity of natural sinks | Many of these ideas are viable but none alone will solve the problem | Not likely to be achieved on a large enough scale cost effectively to make a difference. | ||
| Fertilisation to increase photosynthesis in the oceans: General principles | Increasing photosynthesis in the oceans may have some advantages however there are known and possibly unknown downside risks. | Potential legacies for future generations | Huge costs possibly profitable depending on the price of carbon | |||
| Growing seaweed | Impractical |
The question arises as to what we are going to do with all the seaweed. could it be used as mulch to help get carbon back into desert soils and retain water? | Weed disposal costs and energy may well exceed benefits. | 2 | ||
| Nitrate/phosphate fertilisation | The use of nitrate and phosphate fertilizers would be costly and have huge ecological risks. | The use of nitrate and phosphate would totally upset delicate marine ecologies | Costs may include repair of resulting ecological damage | 2 | ||
| Iron fertilisation | The use of iron fertilizers is relatively cheap but there are as yet unknown downsides. | The full ramifications are not known but research worthwhile. | Costs may include repair of resulting ecological damage | 5 | ||
| Artificial accelerated weathering | Mimics natural weathering process | High cost, significant time lags and possible production of chlorinated hydrocarbon compounds. | Involves the global commons and tinkering with nature. No profitable outputs. | 3 | ||
| Using electricity for growing coral | Desirable but comes with a cost. | Electricity has to be generated. Even if from solar or wind there is still the cost of the infrastructure. | Could be more viable if coral in the shape of building panels was "grown" Sometimes called the "biorock process" | 4 | ||
| Greening the deserts | Desirable. | Always meritorious as can be done on a low cost basis. See our Greening the Desert Movie | N/A | 7 | ||
| Improving Agriculture and Soils | Very desirable as the carbon in soils is an important sink | No down sides. | Improved soils mean greater profits. | 10 | ||
| Planting Trees and Grasses. | Generally a good idea. | The benefit of forests depends on the use of the timber harvested and whether or not old growth forest replaced.Some grasses sequester carbon. See Forest and Grassland Sinks | Mono culture is wiping out species. | 7 | ||
Proposals involving direct disposal of anthropogenic CO2 |
General principles | As a result of the failure of the Kyoto process sequestration is essential | There are many alternatives other than mineral, marine and geosequestration. | Only Gaia Engineering (sequestration of man made carbonate as building material) is uniquely profitable. | ||
| Pumping CO2 gas or liquid into the deep oceans:General principles | Expensive to concentrate and pump CO2 | The downsides are not known. Any up welling of vast amounts of CO2 would be devastatingly lethal. Eventually any CO2 pumped deep underwater would return to circulation. Potential legacies for future generations | Not economically feasible | |||
| Pumping CO2 gas into the deep oceans | Expensive to concentrate and pump CO2 | Acidification is reducing the ability of the oceans to take up CO2. Concentrated CO2 is toxic. Potential legacies for future generations | Not economically feasible | 1 | ||
| Pumping CO2 liquid into the deep oceans | Expensive to concentrate and pump CO2 | The downsides are not known. Any up welling of vast amounts of CO2 would be devastatingly lethal. Eventually any CO2 pumped deep underwater would return to circulation. Potential legacies for future generations | Not economically feasible | 1 | ||
| Geosequestration - Storing CO2 underground | Expensive to concentrate and pump CO2 deep underground | Technologies for concentrating CO2 still unknown. Because of leakage a short term solution at best. May work if CO2 is pumped into highly fractured basic rocks containing water where is would be rapidly carbonated. | Only possibly feasible given a high price for carbon and if used to push up more oil. | 3 | ||
| Mineral sequestration | Involves mining and possibly cartage or pumping | Mineral sequestration will work but neither efficiently nor cheaply | Would cost a lot of money | 3 | ||
| CO2 fixation by algae | Works well enough | Works well enough. Bio reactors are still being developed. | Would cost a lot of money | 6 | ||
| Reacting carbon dioxide with sodium bicarbonate or other minerals | Works well enough | Problem is making the sodium bicarbonate in the first place and then possibly having to get rid of the sodium ion out of material made. | Would cost a lot of money | 2 | ||
| Gaia Engineering - Storing CO2 in our built environment | The use of man made carbonate to build with is uniquely potentially very profitable | There are no known downsides. The technology platform is characterised by profit at every step given economies of scale. Involves TecEco Eco-Cements | Potentially profitable even without a high price on carbon. | 10 | ||
| Gaia Engineering - Using man made carbonate to build with. | The use of man made carbonate to build with is uniquely potentially very profitable | There are no known downsides. The technology platform is characterised by profit at every step given economies of scale. | Potentially profitable even without a high price on carbon. | 10 |
John Harrison our managing director surmised that only a profitable process that converts CO2 and other wastes to resource will work and on this premise his company Tececo have developed Gaia Engineering which is a technology platform that produces building materials from man made wastes including CO2.
Should readers wish to find out more about this wonderful technology which uses man made carbonate to build with then some suggested reading is in the following links:
http://www.tececo.com/sustainability.sequestration_built_environment.php
http://www.tececo.com/politics.analysis.php
http://www.tececo.com/economics.summary.php
http://www.tececo.com/gaiaengineering.synopsis.php
For a tool to compare the TecEco approach to geosequestration go to our tools area
This web page discusses the alternatives most of which we will show just do not stack up in comparison to the TecEco Gaia Engineering solution.
The authors of The Independent article mentioned above have obviously not heard about the TecEco Gaia Engineering technology platform when they reported "that plan B" would of necessity "involve highly controversial proposals to lower global temperatures artificially through daringly ambitious schemes that either reduce sunlight levels by man-made means or take CO2 out of the air. This "geoengineering" approach – including schemes such as fertilising the oceans with iron to stimulate algal blooms – would have been dismissed as a distraction a few years ago but is now being seen by the majority of scientists we (The Independent) surveyed as a viable emergency backup plan that could save the planet from the worst effects of climate change, at least until deep cuts are made in CO2 emissions."
Natural processes have not been able to take up all the carbon produced through human (anthropogenic) activity and we agree that geoengineering (we call it planetary engineering) is in order as there is no doubt "spaceship earth" is in trouble however the solution does not have to involve controversial, wild or wacky and potentially unsafe science. On the contrary it should be part of the way we do things in the future to profitably make a living. Fiddling with the planets homeostasis in a way we may not fully understand is too dangerous to contemplate.
Consider first the fundamentals of any approach to reduce the CO2 in the air:
In nature the photosynthetic process is of major importance
CO2 + H2O + light energy ---> (CH2O)n + O2
Respiration and the burning of fossil fuels on the other hand consumes organic molecules and oxygen to produce energy and CO2.
(CH2O)n + O2 => H2O + CO2
It follows that global atmospheric concentrations of greenhouse gases such as carbon dioxide could be reduced by increasing photosynthesis or decreasing the burning of matter containing carbon (CH2On representing organic carbon and C).
Constraint on emissions is the basis of current efforts however we are too hooked on fossil fuels to realistically reduce emissions. That constraint alone will not work appears obvious to all except perhaps politicians who still compete with one another to see who can promise the greatest reduction. (If you have not already read them please read our economics page and political analysis where we discuss this in detail). The Independent cited previously are right in so far that a focus on technology is required to find ways of evening up the balance. TecEco offer an alternative that will work, that is potentially very profitable and does not have any downside risks. Our solution is to mimic nature and build with man made carbonate. The solution must necessarily involve the way we live and changing what we build with in favour of man made carbonates could uniquely be profitably taken up on the scales required.
CO2 + M => MCO2
Carbonates are the most stable sink for CO2 and over the last 4.5 billion years some 40 million gigatons have been sequestered by nature in this form begging the question whether technologies that mimic this process efficiently thereby providing a viable solution should be mandated by law. Rising to this challenge we have developed Gaia Engineering which involves precipitating man made carbonate from seawater or brine, produces freshwater as a by product and marketable carbonate based building materials.

The fact that there is global warming and it has been caused by "greenhouse gases" including carbon dioxide is not disputed by most scientists. That something must be done is also not an issue and very few, even those who claim there is no long term climate change disagree with the objective of sustainability. What is holding up the process is the practicality. legacy issues and cost.
Kyoto type processes are not the best way and are not proving effective to solve the economic dilemma. There is much economic analyses to date that attempts to show that it will be far more expensive to cut carbon dioxide emissions radically than to pay the costs of mitigating or adapting to the increased temperatures. The argument is that it would be better to spend elsewhere and that for the cost of Kyoto as it is for just one year we could solve the world's biggest problem and provide every person in the world with clean water[4]. Reports from Sir Nicholas Stern[5] and Prof Garnaut[6] and others contradict this however and make it clear that the opposite is true. The costs of not doing anything will be astronomic. All the arguments about doing nothing become fall away in the light of our Gaia Engineering technology as it will pay economically rather than merely legally through an artificial price on carbon so we all undertake activities in such a way so that the problem of global warming is resolved. Other current in vogue sustainability solutions including those in plan B from The Independent and presented by Dr Ben Mathews[1] come at a cost. There is therefore an obvious and urgent need for decision makers to understand and adopt our Gaia Engineering plan which does not involve cost but economic benefit. To get much more out of much less, to produce more with less impact on the greater environment and so on. That more resources are required unless we dramatically reduce our population is difficult to dispute, the need is to produce and consume resources in a sustainable manner by recycling everything as the paradox is that population growth can only be curtailed by affluence. Science got us into the situation and sound science can get us out again. In the case of TecEco some clever science has enabled us to create man made carbonates from calcium and magnesium cations in seawater or brines and not necessarily point source CO2.
TecEco theorised that a good place to sequester carbon was in our own backyards. Our footprint on earth is the built environment and this accounts for around 40% - 50% of all materials flows, emissions and energy consumption. The carbon based TecEco building materials invented by John Harrison our MD use man made carbonate aggregates as well as binders and both are made with no releases. The result is massive potential sequestration driven by profitable sales into a huge continuing insatiable market.
The recent economic downturn has furthermore demonstrated that it is difficult to think about the effect of our actions on the environment when we are forced to focus on remaining economically viable. A way forward must involve profitably achieving a solution to the most pressing problem ever to face civilisation and Gaia Engineering represents this opportunity.
To highlight the advantages of the Gaia Engineering technology platform other plan B alternatives including those mentioned by both The Independent and presented by Dr Ben Mathews[1] are described below with commentary.
It goes without saying that the more efficient the use of energy can become, the less is required for a given outcome. Any advances to efficiency of processes also save money in that the consumption of less energy to achieve a given outcome means lower input costs and a better bottom line. Unfortunately increasing energy efficiencies are reaching the law of diminishing returns and besides there are limits as Di Fazio explains [16].
Renewable energy is energy not from fossil fuel sources and involves capturing current everyday energy from the sun such as manifested in solar, wind and tidal power.
The more renewable energy can replace the use of fossil fuels the better. The missing factor is technology which can hopefully make such renewable sources of energy competitive and able to meet base load demands. The technology platform defines what is or is not a resource (See Pilzer's law)
Few people realise it but we have been controlling the climate for thousands of years[7]. What we have to do now is do so deliberately if civilisation is to survive.
According to Dr Ben Matthews[1] original article current technical proposals fall into three main categories: increasing the reflection of solar radiation back to space, enhancing natural sinks of carbon dioxide, and direct disposal of carbon dioxide captured at source. The discussion below follows these headings.
The temperature at the Earth's surface slowly adjusts so that the energy from incoming solar radiation (sunlight- ultraviolet and visible) is balanced by terrestrial radiation (infra red) emitted from the Earth. When greenhouse gases reflect back some of that terrestrial radiation, the surface warms and emits more radiation, until the amount escaping the atmosphere balances the sunlight as before.
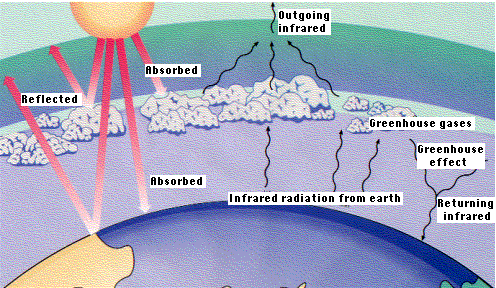
To offset the warming effect of the predicted rise in greenhouse gases in the atmosphere it has been suggested that we could reduce the incoming solar radiation by intercepting about 1% of sunlight.
Giant reflectors in space, and stratospheric dust or aerosols are two such methods proposed. There are however a number of problems with these schemes discussed below.
According to the Independent[2] "Volcanic eruptions release huge amounts of sulphate particles into the upper atmosphere, where they reflect sunlight. After Mount Pinatubo erupted in 1991, sulfates reflected enough sunlight to cool the Earth by 0.5C for a year or two. The Nobel Laureate Paul Crutzen suggested in 2006 that it may be possible to inject artificial sulphate particles into the upper atmosphere – the stratosphere. However, the idea does not address ocean acidification caused by rising CO2 levels. There may be side-effects such as acid rain and adverse effects on agriculture"
Another idea has been to modify jet airplane fuel to do this. Besides being cheap, the aerosol fix is also promoted as "reversible", i.e. it can easily be stopped if it doesn't work, and within a few years the dust would fall out. Most greenhouse gases have a much longer lifetime so if they are to be offset with stratospheric aerosols, we would have to rely on the ability of future generations to keep flying those planes, to keep repairing the shield or be faced with sudden warming. There would be questions as to the cost and supply of suitable chemicals. Even if we are content to pass on these burdens, we would also be cutting the amount of sunlight reaching plants on the surface, and presumably also changing its spectral composition. Perhaps the plants would take up less CO2? And do we really want to live under a constant haze in the sky to keep us cool? Do a few scientists and policymakers have the right to impose this on all other life on the planet? Do we want to breathe the chemicals? Another obvious objection is that the injected particles might provide a very efficient surface for ozone destruction, as polar stratospheric clouds already do every spring.
Again according to the Independent[2] "Some scientists suggest it would be possible to deflect sunlight with a giant mirror or a fleet of small mirrors between the Earth and the Sun. The scheme would be costly and prompt debate over who controls it. Many scientists see it as contrary to the idea of working with the Earth's systems." This idea is to put gigantic foil sheets up in orbit around the earth to reflect sunlight. They would periodically cast a shadow, intercepting incoming light about 1% of the time, and would be assembled in space because such things couldn't be launched from down here. Sound a bit wacky? We think the idea is very wacky. The sky would rain bits and pieces of reflector all the time as they broke up.
Another idea is to increase the Earth's albedo, or reflectivity to sunlight, by pumping water vapour into the air to stimulate cloud formation over the sea. The Independent state that "John Latham of the United States National Centre for Atmospheric Research in Boulder, Colorado is working with Stephen Salter of Edinburgh University and Mike Smith at Leeds to atomise seawater to produce tiny droplets to form low-level maritime clouds that cover part of the oceanic surface. The only raw material is seawater and the process can be quickly turned off. The cloud cover would only affect the oceans, but still lower global temperatures." Again there are numerous potential downsides. The salty clouds my drift over agricultural land and ruin it and then who would pay the bill? Besides the effect of such costly schemes would be small compared to natural feedbacks.
The idea is to enhance natural sinks such as photosynthesis, growing coral or weathering. Many of these ideas will work and some such as greening the desert have other obvious advantages. Some however such as fertilising the oceans carry unknown downside risks.
The principle nutrients limiting the growth of ocean algae are nitrate, phosphate, and in some places, iron. To some extent we are already increasing the nutrient supply to coastal waters, through agricultural and sewage runoff. However, further from shore the nitrate and phosphate are supplied mainly by up welling of deep water. Over much of the ocean, where warm surface water rests stably above cold deep water, the nutrient supply is poor and little grows.
There have been many proposals to grow more algae by augmenting the nutrient supply. Not only might this enhance the biological carbon pump, but it might also provide a fuel or even food for fish.
On the other hand, our ability to predict the many feedbacks intrinsic to marine ecology is still very poor as the role of algae and bacteria in recycling nutrients is only just beginning to be uncovered.
At least in surface waters the various perturbation effects could be easily measured. Once the surplus algae sank deeper however there is little consensus as to their fate. One concern is that the extra supply of organic carbon could use up all the oxygen in parts of the deep ocean and this might lead to the production of a lot of methane, or nitrous oxide which are much more potent greenhouse gases than CO2. Plankton also produce other gases which affect climate such as dimethyl sulphide which oxidises to form sulphate aerosols which seed clouds (see above), and smaller quantities of hydrocarbons and halocarbons which also affect atmospheric chemistry.
There has also been little consideration of the effect on any life in the sea bigger than algae. As a general rule, biodiversity in the sea decreases in highly productive algal blooms. And suppose we really could predict the effect on all the plankton, krill, fish, whales etc.? Marine ecology is a complex non-linear system that behave chaotically and science is far from making such predictions. Consider, for example, the failure of the scientific management of fish stocks, or the poor understanding of toxic algal blooms.
Seaweed dominated the early ocean-algae climate engineering proposals. The idea was to set up kelp farms, eventually covering tens of thousands of square kilometres of the open ocean, originally with the intention of producing methane. In the 1970s $20 million of research was funded by the (then) American Gas Association, only to find that it would cost them 6 times more than the energy they would gain. The vast cost comes from the need to supply nitrate and phosphate to the surface ocean. Either you can add man-made chemicals, in amounts well exceeding the total world production of fertiliser, or use a lot of energy to pump up nutrient rich water from the sea floor. It has also been pointed out that such water is usually supersaturated in CO2, which would then be released to the atmosphere.
In the heat of the greenhouse effect and with Carbon taxes/credits in sight, the seaweed idea has been revived, mainly by the US Electric Power Research Institute. They say it would cost about $200/tonne C sequestered. Engineers have devised grand schemes with diagrams showing the tracks of supertankers moving about the farms harvesting the seaweed. What they are going to do with it is still not certain.
Both the European Community and the Japanese have recently supported research investigating coastal fertilisation to increase both the biological carbon sink, and the supply of fish. Such projects are simplistic and will probably do far more harm than good such as encouraging jellyfish, anoxia or toxic algal blooms. Apparently they have found some commercial support such as from Norsk Hydro (one of the world's biggest manufacturer of fertilisers) a company who wish to add nitrate and phosphate to the Norwegian sea and that are already experimenting in fjords. Meanwhile Mitsubishi are funding a similar proposal off Japan, claiming it might not only capture carbon dioxide, but also produce a lot of sardines.
Jones (1996)[8] calculates the nitrogen needed to soak up the entire projected anthropogenic global CO2 emissions and claims to get 260 kilos of fish per tonne of nitrogen.
Of all climate engineering proposals, fertilisation of the Southern Ocean with iron has raised the most controversy. Perhaps this is because experiments designed to investigate whether iron is the key limiting nutrient are already underway and well publicised.
The idea has been around for about 20 years, and is credited to John Martin who first developed the clean laboratory techniques to measure how little iron there was dissolved in open seawater. The concentration is low because it falls out as a precipitate from alkaline seawater, so the only supply to the open ocean is atmospheric dust. This led Martin to suggest that iron might be the limiting nutrient, which would explain an old puzzle: Why, in the Southern Ocean and the Equatorial Pacific, is the algal growth much less than would be expected from the supply of nitrate and phosphate? If iron was the answer, as suggested by bottle incubations of the algae in these waters, it might also be a feedback controlling ice ages. Atmospheric dust increases during glacial periods. This dust could fertilise the Southern Ocean and the algae would soak up enough CO2 to reduce the greenhouse effect, enhancing the ice age.
These considerations led to John Martin's famous quote at a conference, "give me half a tanker of Iron, and I'll give you an ice age". It was a joke. But once this idea was out, for the "biggest manipulation of nature ever attempted by man", it seemed inevitable that scientists would want to try it out, albeit on a small scale. The proposal briefly caught the attention of the US media, with portrayals of irresponsible mad scientists in white coats about to take over the world. It was feared especially that the possibility of a technofix to global warming would weaken resolve to reduce CO2 emissions. To calm the uproar the scientists adopted a resolution, including "The American Society of Limnology and Oceanography urges all governments to regard the role of iron in marine productivity as an area for further research and not to consider iron fertilisation as a policy option that significantly changes the need to reduce emissions of CO2".
Prof Andy Watson does not rule out the possibility that iron fertilisation could one day lead to a climate engineering fix ."We are interested in the possibility that something as relatively simple as this could be used." But his model predicts that even a continuous widespread iron fertilisation could only reduce atmospheric CO2 by 60ppm by 2100, a tenth of that needed to offset "Business-As-Usual" fossil fuel emissions.
Like Gaia Engineering in its full implementation iron fertilisation is cheap, perhaps as low as 5$ per tonne of Carbon fixed, compared to much higher figures for other proposed sinks. Iron needs to be added continuously, but not too much. Aeroplanes could deliver dust, or rusting tankers or discarded oil rigs could produce it in situ. A more sophisticated approach might employ purpose-designed slow-release floating granules.
Scientists are however far from being able to predict the effect on the ecology, as already noted in the general points on ocean fertilisation (above). According to The Independent " Experiments support the idea, but blooms may be eaten by animals so carbon returns to the atmosphere as CO2." It has also been suggested, that iron fertilisation could alter the dynamics of the Southern Ocean to increase the natural flux of iron-rich water from depths to the surface, resulting in a runaway iron fertilisation. This physical feedback might be initiated by a decrease in sunlight penetration through the algae. So it is quite possible, that we could underestimate the feedbacks and go too far, thereby creating another ice age.
It is not surprising that iron fertilisation has raised so much controversy. Of all climate engineering proposals, it is perhaps the easiest and cheapest to carry out, very elegant, and yet it carries many unpredictable consequences. It also involves the pristine ocean around Antarctica, the part of the world least affected by our pollution so far. Experiments have already been "successful", and the organisers need to publicise the results to help secure more funding, for they have so far avoided commercial sponsorship and rely on government research funds. Perhaps because it is investigated openly by independent ocean scientists, the idea retains more credibility than it would if backed by industry. Some claim that we should push ahead with research, to ensure the results are open for the world to judge, before any commercial venture can get established. There is already an international race among oceanographers to get the money for the next experiment. But the Southern Ocean belongs to none of us, perhaps more rightly to whales, krill, penguins, algae, all life on earth. Respect for this seems to have been lost in the race to be first with clever science.
We think the research should be on trying to determine to what extent iron fertilization was a feedback mechanism that worked to reverse ice ages. In this sense the solution mimics what has happened in nature albeit in the past. The fertilzation of the oceans is covered in more detail in Newsletter 74
In this process promoted by Harvard and Pennsylvania State University[9] electrochemically produced hydrochloric acid is extracted from seawater using a modified chloralkali process and neutralised by artificially weathering mafic rocks thereby increasing the total alkalinity of seawater and hence its ability to absorb more CO2.
The process would have to be adopted on an incredibly large scale to make a difference and has no economic drivers other than the price of carbon. Once again it is fiddling with the global commons with unknown downsides including the amount of chlorinated hydrocarbon compounds that may also be produced. We think this process may be able to be coupled with the Greensols process giving it some economic drive.
If a low voltage DC current is passed through a metal grid acting as a cathode, calcium carbonate and magnesium hydroxide crystals will grow on it encouraging the formation of coral. The anode is usually a titanium mesh that does not corrode. Solar, wind wave or tidal power could act as the energy source and the lower the current the safer for fish. Coral larvae attached to the carbonate surfaces and grow rapidly.
This process seems to work well for reef regeneration after storms and tsunamis and has been adapted to grow coral walls and other building components.
People have always dreamed of greening the world's deserts. Their potential as a CO2 sink as well as a food source has revived interest in such grand schemes. One such proposal from the Japan Gas Association and RITE includes generation of clouds by evapotranspiration from coastal mangroves and lagoons, and artificial mountains to promote rainfall, along with underground dams and new cities. They even provide an "artists impression" of this new landscape.
A slightly less ambitious and more realistic scheme involves using halophytes. These are plants, usually found in salt marshes, that thrive in saline conditions. The idea is that, perhaps with a bit of genetic engineering, they could be adapted to desert lands irrigated with seawater, or lake/river water that has become too salty for any other purpose. Glenn estimated that world-wide there are 130 million hectares of suitable land, and that this could sequester about 0.7 billion tonnes Carbon annually, at a cost of about $200/tonne. It is suggested that some of the crop could be ploughed back into the "soil", the rest could be buried dry. He also recommends a particular oilseed crop, that is edible, tasty, nutritious, and could also be a fuel. The main problem is that you need a lot of excess irrigation to leach out the salts that would otherwise build up. Presumably you also leach out nutrients at the same time, so where do you replace them from? These questions are not addressed properly and we recommend reading our web page on the salt problem and the water cycle
Some other obvious points seem to have been overlooked. If you make the desert wet (or even just grow trees on it), it becomes darker, thus absorbing considerably more sunlight and warming the planet. There would also be very high evaporation, and water vapour itself is a greenhouse gas. On the other hand, if more clouds formed as a result, they reflect sunlight. We should also recall, that rice paddy fields are a very large source of methane, and these salt marshes might be likewise.
We think the way to go is using combinations of swales, mulch and appropriate plantings and invite readers to view the flash movie on our web site about permaculture from the ABC, Australia's public broadcaster.
Around 50 – 80% of the organic carbon that was once in the topsoil has been lost to the atmosphere over the last 150 years or so due to our failure to take care of the earth as a living thing. By inference, degraded soils have the potential to store up to 5 times more organic carbon in their surface layers than they currently hold, provided we change the way we manage the land. Appropriately managed soils will sequester atmospheric carbon and the world's soils already hold two or three times as much carbon as the green matter above them. In the humification process that occurs in well managed soils soil carbon increases and there are significant other benefits including fertility because of pH buffering, inactivation of pesticides and pollutants, improved plant nutrition and increased soil water holding capacity.
Carbon in soils is so important we have devoted a web page to it. See The Role of Soils in Sequestration. Putting carbon back into soils is common sense and very important if we are to prevent runaway global warming.
There are plenty of good reasons to plant trees, and carbon storage is only one of them. Although reforestation might be considered climate engineering, it takes place within national boundaries rather than exploiting the "global commons", and also it could hardly be considered a new unknown technology! Trees can be planted by people locally and they know what to expect as a result.
There are a few common misconceptions. For instance, mature forests do not take up much carbon, only young forests are net sinks. As a forest matures, it approaches equilibrium where growth nearly equals decay. As a consequence many do not consider forestry as a long term solution only if wood is continuously harvested and stored. It has also been suggested that we fertilise existing forests to maximise carbon uptake, this would likewise provide only a temporary sink.
On the other hand, some forests and grasslands and peat bogs in particular, can be a permanent sink for carbon in the longer term, as more accumulates on top each year and ends up in the soil. Recent reports suggest that carbon sinks formed in this way may be equal or greater in magnitude than the world's trees. According to the figures presented by Ziok depicted in the diagram above plants account for some six or seven thousand gigatons and carbon in soil for a little over double that.
It is not always wise, from a climate perspective, to put trees where there were none recently before as carbon is generally lost from the soil. If a peat bog for example is dried out by planting trees, the previously anaerobic peat becomes accessible to soil microorganisms, which release it as CO2, or worse, as methane. Generally however planting trees is a positive thing to do to combat global warming and climate change.
For further reading see "Tree Farms Won't Halt Climate Change" in Newsletter 25 and "The Role of Nature in Sequestration Given Geological Time Scales" in Newsletter 21.
We don't think that forests should be planted as a replacement for existing old growth forests or mature grasslands. Rather marginal land should be recovered with them (See Forests and Grasslands as Sinks)
Direct disposal attracts funding because a company can then dispose of its own CO2 and thereby avoid taxes or emissions quotas and as a consequence various proposals have had the most publicity. UNfortunately some of them are driven by ulterior motive such as to push oil up in the case of geosequestration underground. The two main methods so far advanced which are oceanic and geosequestration involve the "global commons" rather than the property of any particular company or government and we all have a right to be concerned. The disposal of CO2 into the global commons raises many ethical and scientific dilemmas Furthermore all the proposals other than Gaia engineering from TecEco require point source CO2 and as yet there are no economically viable methods of concentrating the gas.
The deep ocean has an enormous capacity and is a natural medium-term sink for carbon however we believe the best long-term sink is not in seawater, not geosequestration but fixed as carbonate building materials.
The deep ocean has an enormous capacity to store carbon dioxide. This is principally due to the pH of seawater, such that for every 100 molecules of CO2 stored in it, roughly 98 of these are found as bicarbonate ions, 1 has been converted further to a carbonate ion, and only one remains as CO2.

The problem is that most of the ocean water is not in contact with the atmosphere. Transfer of CO2 between the surface ocean and the deep ocean is slow, and occurs by two main processes: subduction of cold salty waters, particularly in the North Atlantic , and the biological "pump" whereby organic particles sink below the mixed surface layer.
Only a small fraction of these particles eventually reach the sediment, the longest-term sink of carbon[3] , but on the timescale of a thousand years or so removal to the deep water itself is sufficient. If this happened faster, the pulse of CO2 in the atmosphere from fossil fuel burning would be much smaller, and thus the greenhouse warming less dramatic, although the long-term equilibrium climate change would be unaffected.
Climate engineers want to get CO2 into the deep ocean faster. There are various proposals: CO2 could be pumped there directly, soaked up by changing the alkalinity of the water, or absorbed into an enhanced biological pump by "fertilisation" of the ocean with nutrients discussed above.
Surface seawater is also typically supersaturated with respect to calcium carbonate, and it might be expected that precipitation of this solid (directly, or into shells or corals etc.) would be a good way to remove CO2 from the system. Paradoxically, it actually does the opposite through acidification. While ocean chemists have long understood this, it seems that some engineering consultants have yet to work it out, proposing, for example, to increase the growth of calcareous seaweeds.
Another related effect of the seawater carbonate chemistry is also often ignored. For every ten units of CO2 removed from the surface ocean, about 9 are replaced from the vast pool of bicarbonate ions in the water, and only about 1 would be replaced from the atmosphere. So if, for instance, seaweed was used as a fuel, the main effect would be to shift CO2 temporarily from the ocean to the atmosphere. It may be better than burning fossil fuel, but is not an efficient way to solve the problem.
According to The Independent "The Earth scientist James Lovelock, working with Chris Rapley of the Science Museum in London, devised a plan to put giant tubes into the seas to take surface water rich in dissolved CO2 to lower depths where it will not surface. The idea is to take CO2 out of the short-term carbon cycle, cutting the gas in the atmosphere. Critics say it may bring carbon locked away in the deep ocean to the surface." Much as we have considerable respect for James Lovelock, this idea is expensively wacky.
Injecting CO2 as a gas into the deep oceans would have disastrous effects on sea life as a result of acidification and toxicity. Decreases in the pH levels of seawater would result in dramatic physiological effects on many species. Shallow-living organisms like shelled mollusks and corals are already being affected by the growing levels of CO2 in the atmosphere which are causing acidification and inhibiting the ability of shellfish to form shells and causing coral reefs to dissolve. Deep-ocean sequestration would involve concentrating an otherwise dilute toxin to well above lethal levels, and placing it in an environment where the organisms are not tolerant of environmental fluctuation. Localized devastation of biological communities at the injection sites is certain.
The gas would first have to be liquefied, and then pumped down pipelines (for which the technology is still being developed) to below 1500m depth (for environmental reasons -see below). It would then mix with seawater, forming a very acidic plume which would spread out across the sea floor.
Originally it was thought 3000m was necessary, because below this depth the pressure is sufficient to keep CO2 as a liquid. However, various groups then claimed shallow injection was possible because, so long as you could get enough CO2 to dissolve before the bubbles rose to the air, then the resulting dense CO2 solution in seawater would sink naturally. They looked for sites where ocean currents already descended continental slopes.
A complicating factor is that when concentrated CO2 and water are mixed, they react to form solid compounds known as clathrates. In trial experiments, the clathrates blocked the end of the CO2 pipe. A lot of research then followed on this topic and it was even proposed that the clathrates may be useful, because they sink rapidly helping to solve the depth problem. The Japanese are particularly keen on the idea of deliberate clathrate formation as clathrates are being considered as a fuel.
When CO2 dissolves in water it forms hydrogen carbonate and carbonic acid which could kill most marine life, perhaps over a large area of the sea floor. Usually the chemistry of the benthic environment changes very little, so even a small perturbation may have disastrous effects[1]. Perhaps the engineers view the deep sea as worthless mud with a few worms in it. However, marine ecologists have recently estimated that there are so many different species of benthic organisms, that the biodiversity is comparable to the tropical rainforests. We just don't know much about it yet so does this give us a right to destroy such life to satisfy our thirst for burning oil? What affects would doing so have on the food chain?
A few of these questions have been considered and at the 1996 International Energy Agency Workshop on environmental impacts[11] the marine biologists present came up with some fairly restrictive criteria, such as "no species should be driven to extinction" and "no significant destruction of ecological processes at basin scales", for which there must be no acidic strata which could form a barrier to migration. To be sure of that, and to protect diversity of shelf slopes, CO2 should be released below 1500m. On the other hand, a pure "CO2 lake" sitting on the sea bed (it's denser than water) would be disastrous for life in the sediment.
The high concentration of CO2 in the seawater would also reduce its capacity to hold other gases such as oxygen and the bottom water might therefore become anoxic. Methane could then form, although proponents assert it wouldn't rise to the sea surface. We do not believe this as deep ocean currents change over time, sometimes suddenly, rarely predictably and always depending on finely balanced physics. Instead of welling slowly to the surface, the CO2-rich water might become unstable while the CO2 is still concentrated, rising suddenly as plumes of gas as happened in 1989 at lake Nyos in Cameroon. Both methane and CO2 do this and there is a theory that such events may account for the mysterious loss of ships in the "Bermuda triangle". Nobody can explain these sudden gas destabilisations and pumping liquid CO2 to the bottom of the sea, could lead to similar disasters. A sudden large pulse of CO2 from ocean storage up welling could cause enough warming to trigger climate feedback mechanisms that lead to a runaway greenhouse effect. given this scenario it would have been much better to have put the CO2 into the atmosphere, year by year as it was produced!
Another problem with oceanic storage is that it takes a lot of energy to pump anything down to such a pressure under the ocean, and a lot more fossil fuel has to be burned to make this extra energy, so this process is extremely inefficient. There seems to be some disagreement in the literature as to exactly how much more energy is needed, but it is at least 30-40% extra. Part of the confusion arises because both CO2 "capture" from flue gases (essentially an entropy problem) and CO2 "disposal" (transport, pressurising) cost energy, but where one stops and the other begins is arbitrary. Furthermore the costs are much higher for conventional power stations than for new ones purpose-designed for CO2 capture.
Not only will the additional energy cost more, in the long term it will also make the CO2 problem worse. The unsatisfactory result is that more CO2 (eg 40%) has to be disposed of into the deep ocean than would have gone into the atmosphere if it had gone all gone up in smoke as is currently occurring. Over hundreds of years through ocean circulation and diffusion, the CO2, including that "extra" CO2, will find its way back into equilibrium with the atmosphere further compounding the problem putting an extra burden on future generations in order to avoid a problem now. This is an issue of intergenerational equity, which isn't usually noticed in any cost-benefit analyses because the future is so rapidly discounted.
As the global climate warms and CO2 increases in the atmosphere, the deep ocean's buffering capacity for CO2 uptake also decreases and becomes a less favorable option.
Although the ocean is a much bigger sink, some CO2 can feasibly be stored underground in aquifers or depleted oil and gas wells. Such projects are already underway both in Texas, below the North Sea from a Norwegian platform and in Australia. For this technology to succeed it is important that there is a very low leakage rate and given the fractured nature of the trust and seismic activity this is unlikely. We think that the only scenario whereby geosequestration may work would be if CO2 is pumped into highly fractured basic rocks containing water where is would carbonate. Otherwise we think it too dangerous.
Geosequestration has become the coal industries answer to the carbon dilemma with the catchphrase "clean coal". The idea is to pump CO2 underground (usually with the co-operation of the oil industry as the gas is used to force up more oil.) Our comment is that much work over many years during the cold war and later has established that the risks of pumping CO2 underground are far too high due to the fractured unstable nature of the crust. This risk the oil industry are keen to ignore and transfer to future generations.
The Climate Action Network Australia (CANA) which is an alliance of environmental, public health, social justice and research organisations working together to fight Global Warming produced an interesting graph in a 2004 media briefing which supports our own modeling of geosequestration (downloadable under tools). The models vary depending on the emissions and response scenarios adopted but importantly they concurrently prove that with even with small amount of leakage which will be inevitable, that it is essential we adopt a permanent sequestration technology such as Gaia Engineering.
How effective goesequestration is depends mainly on the leakage rate which of course can never be determined - the crust is instable in the long term and that is why we have the continents we do and so on. As a consequence researchers vary greatly in their assumptions and conclusions regarding a likely level of leakage. Whatever the scenario undeniably some leakage will occur because geological formations are not completely stable. They are for example disturbed by earthquakes caused by the movement of plates or up welling of magmas. Another source of leakage could also be the instability of injection points over time[12].
Dooley and Wise[13], Hawkins[14], and Hepple and Benson[15] find that the annual leakage rate must be lower than 0.1% if geological storage is to be safe. The problem is that if the leakage rate is higher, targets for atmospheric greenhouse gas stabilization in the range of 450 to 550 ppmv become unattainable in the long term. Our own modeling in our new downloadable Gaia Engineering Process v Geosequestration Model under tools indicates that anything more than about .2% leakage which is quite possible would result in the problem getting worse after about 200 years and at best all geosequestration can do is buy us time.
The diagram below from CANA [16] illustrates a scenario in which geosequestration is used as the exclusive greenhouse gas emission reducing tool for the next two hundred years and a leakage rate of 0.1% per annum is assumed. The graph shows that by the end of the 22nd century the entire ‘carbon budget’ of future generations would be consumed by leakage from past underground carbon dioxide storage. This would mean that future generations can not avoid dangerous climate change, even if they reduced their own greenhouse gas emissions to zero. Geosequestration like every other form of direct disposal of anthropogenic CO2 discussed so far merely transfers the problem to future generations.

In conclusion CANA said that "The assumption of exclusive reliance on storage may be an extreme one, however the example illustrates that emphasis on energy efficiency and increased reliance on renewable energy must be priority areas for greenhouse gas mitigation. The higher the expected leakage rate and the larger the uncertainty, the less attractive geosequestration is compared to other mitigation alternatives such as shifting to renewable energy sources, and improved efficiency in production and consumption of energy."
TecEco believe the graph demonstrates that a permanent form of sequestration is essential because of the leakage issue. Our view is that it will be too difficult given human nature for us to give up the remaining 7,000 gigatons of coal left on the planet as a source of energy and that the rate of conversion to non fossil sources of energy cannot possibly be quick enough. As Di Fazio has demonstrated - the correlation is just too strong [17] between world industrial product (WIP) and fossil fuel energy and thus emissions. Gaia Engineering involving the production of man made carbonate building materials is the most promising form of permanent sequestration yet devised. Logically there is no other choice.
Geosequestration is still too expensive for large-scale operation unless there is some other advantage such as forcing up more oil. In current installations it comes from under the rocks in the first place, mixed with the oil and gas deposits. Gas from the Sleipner Vest gas field off Norway contains 9.5% CO2, most of which has to be separated from the methane before it can be sold. The recent introduction of a carbon tax (180$/t C) in Norway encouraged Statoil to set up an installation to pump the CO2 (about one million tonnes a year) into a sandstone aquifer 1000m under the platform[18]. This is seen as a pilot project by the promoters for burial of up to 1/3 of all Europe's CO2 emissions.
To bury 1/3 of Europe's CO2 emissions clearly requires much more than separation of the CO2 initially mixed with natural gas. Even now separating CO2 from other flue gases at conventional power stations is very expensive and inefficient.
As an alternative companies like Statoil envisages a "hydrogen" economy whereby the fuel is converted into CO2 and H2 rather than being oxidised completely in combustion (Kaarstad 1995). The hydrogen (note it's highly explosive!) is then used to power transport whilst the CO2 is buried.
We mention leakage as being the main issue and this could be sudden and catastrophic. If CO2 stored for several decades suddenly re emerged as a sudden pulse to the atmosphere, the resulting sudden greenhouse warming could be catastrophic. On the other hand, perhaps the rocks are slightly more secure than the deep ocean, for which the same applies. As for pumping CO2 down into the deep ocean considerable energy is required reducing the efficiency of the process and producing CO2. Another concern is that the CO2 could contaminate groundwater in nearby aquifers, making it acidic and unsuitable for many purposes.
Even if these concerns could be met, it is unlikely that enough suitable storage/carbonate conversion locations are available to remove a large fraction of world CO2 emissions.
TecEco cover the geosequestration issue in detail in Newsletters 51, 61 and 64, 69 79 and 81 (see Newsletter Back Issues) as well as in our as well as in our economics and political sections. See also our submission to the Australian Parliaments Committee of Enquiry into Geosequestration. We also have prepared a computer model comparing our Gaia engineering technology to sequestration downloadable from our Tools area.
Mineral sequestration is a term generally used in reference to using serpentines, olivines or peridotites to sequester CO2. The technology was first mentioned by Seifritz (Seifritz 1990 [19]) and discussed further by Dumsmore (Dunsmore 1992 [20]). However, Lackner and his associates (Lackner, Wendt et al. 1995 [21] ) were the first to provide the details and foundation for current research into the technology.
Permanent sequestration as a solid stable carbonate mineral is axiomatically much safer than pumping CO2 as a liquid or gas into underground reservoirs and to this extent support mineral geosequestration as suggested by the above authors and others.
The main problem with the abovementioned technologies however the high cost which are furthermore not offset by the production of useful by-product.
More details are to be found in Newsletter 69 and our submission to the Australian Parliaments Committee of Enquiry into Geosequestration
Another seriously considered idea, for on-site treatment of waste CO2, brings us back to algae again. Algae comprise a vast group of photosynthetic organisms, which have an extraordinary potential for cultivation. They can be cultivated under difficult agro-climatic conditions and are able to produce a wide range of commercially interesting compounds such as hydrogen, fats, oils, sugars, carbohydrates and functional bio active compounds from water and CO2 using light as a source of energy. Some strains of algae are for example effective for the manufacture of hydrocarbons that are suitable for direct use as high-energy liquid fuels. Alcohols can be made from cellulose produced by many other algae and the hunt is on for suitable strains and genetic material. Producing fuel from carbon dioxide by using photosynthetic algae is important not only because the process will offset the decline in oil, but because it also attracts carbon credits and insulates against oil price rises. With many other resources starting to run out substitute timbers, papers, fabrics chemicals and foodstuffs that algae can begin the supply chain for are of interest.
Farming algae in bioreactors, tanks or lakes is really a form of solar power, whereby photosynthesis converts CO2 back into organic carbon and that is why such large areas or special light delivery systems are required. Various attempts are being made to culture and genetically engineer algae specifically for this purpose and we discuss this option in Newsletter 64.
This reaction is well known and could obviously be used as the basis of creating solid material made of hydrated magnesium carbonates i.e. a concrete. Using a modified chloralkali process salt water may be able to be split into its acid and alkali components. The main alkali produced is sodium hydroxide and this could be used to absorb CO2 and carbon credits claimed. Although the CO2 could then be shifted to magnesium by reacting sodium carbonate with magnesium oxide and water this is not the sequestration step and magnesium oxide would still have to be made which would unless produced in a closed system as in our Tec-Kiln have further releases.
Our conclusion is that it would be more economic to couple a modified chloralkali process with the Greensols process and produce carbonate in this manner. The alkali produced would qualify directly for carbon credits because it would increase the total alkalinity of the oceans and thereby its ability to absorb CO2 and the acid produced could be used in the Greensols process. As carbon credits would ensue from both the acid and the alkali routes such combined processes would be more economic that the accelerated weathering process previously mentioned and trying to make a concrete directly from sodium carbonate.
Figures abound as to the size of our built environment but are in broad agreement. According to the Australian Federal department of Industry Science and Tourism[22] buildings are responsible for some 30 % of the raw materials we use, 42 % of the energy, 25% of water used, 12% of land use, 40% of atmospheric emissions, 20% of water effluents, 25% of solid waste and 13% of other releases.
TecEco Pty. Ltd. have patented the addition of reactive magnesia to other hydraulic cements including Portland cement in virtually any proportion. At one end of the scale, with high proportions of Portland cement (modified Portland cements) is higher strength but lower sustainability – at the other end of the scale in permeable substrates Eco-Cements carbonate at a reasonable rate with Portland cement which provides the nucleation sites for carbonation in an open 3D lattice as the catalyst. There is also an increased capacity to utilise wastes of all kinds. All formulations are more durable. Because Eco-Cement concretes are substantially recyclable, low energy, at worst nearly CO2 neutral and at best, when man made carbonate aggregates are also used and both the aggregates and Eco-Cements are made without release - huge carbon sinks. If widely deployed the impact on global warming is sufficient to reverse the problem.
The sequestration that would result from the inclusion of man made carbonate as aggregates as in the Gaia Engineering paradigm or even waste carbon based matter including sawdust, waste timber shavings and chippings, plastics, rubbers etc, many of which are currently burnt adding to the global atmospheric CO2 level, would be colossal. The amount of sequestration that would result by including waste carbon based materials is unknown but also large. Many of these materials will add desirable properties such as strength or low conductance to Eco-Cement man made carbonate products
CO2 is a greenhouse gas which traps infra red radiation and concentrations are higher than they have been for 20 million years and some 30% higher than before the industrial revolution. According to the IPCC these increases in CO2 levels probably explain the +.6 +-.2 temperature increases that have occurred in the last century[23]
The earth is a complex homeostatic system[24] and regardless to the extend to which anthropogenic increases in carbon dioxide levels are contributing to global warming and climate change we should not be altering the system.
Desirable as it is, a reduction in the use of fossil fuels seems unlikely as was first recognised by Vernadsky in 1924[25], by di Fazio many years later[17] and explained by John Harrison on TecEco's economics and political analysis pages.
This web pages describes ways of increasing the biological sink from the feasible to the absurd. Leaving old growth forests alone and growing new ones makes sense as does greening the desert. Improving soils so they retain carbon makes even more sense. Less feasible and more risky especially in the longer term are plans to stimulate the ocean biological pump by adding missing nutrients such as iron.
Injecting CO2 into the deep ocean carries with it even higher risk as does injection into geological strata except perhaps where reaction with alkaline results in secondary carbonation formation.
Another popular solution, mineral carbonation, although safe does not carry with it any potential for profit and given the parlous state of global economies is therefore unlikely to proceed.
All of the above cannot possible be achieved on sufficient scales quickly enough to solve the problem as the present excess flux of CO2 is around 8-9 gigatons And some carry unacceptable legacy risks
Not surprisingly the solutions are under our control. We are responsible for the loss of carbon from soils and can put it back again. We can build with the very same carbon we emit from our exhausts. By changing our agricultural practices and the materials we use in favour of man made carbonates to create the built environment there would be sufficient flows of carbon back to soils and more permanent carbonate sinks to profitably solve the problem.
[1] Mathews, B. (1996). Climate Engineering, A critical review of proposals, their scientific and political context, and possible impacts.compiled for Scientists For Global Responsibility.
[2] Conner, Steve and Green, Chris, The Independent, Friday 2nd January 2009 at http://www.independent.co.uk/environment/climate-change/climate-scientists-its-time-for-plan-b-1221092.html
[3] Ziock, H. J. and D. P. Harrison. "Zero Emission Coal Power, a New Concept." from http://www.netl.doe.gov/publications/proceedings/01/carbon_seq/2b2.pdf.
[4] Lomborg, B. (2002). Viewpoint: End Global Poverty Before Global Warming. National Geographic News and many other sources.
[5] Stern, S. N. (2007). "The Stern Review on the economics of climate change." Retrieved 05 March 2007, from http://www.hm-treasury.gov.uk/independent_reviews/stern_review_economics_climate_change/stern_review_report.cfm.
[6] The Australian Garnaut Review Final Report at http://www.garnautreview.org.au/domino/Web_Notes/Garnaut/garnautweb.nsf
[7] Ruddiman, W. F. (2007). Plows, Plagues, and Petroleum: How Humans Took Control of Climate. Princeton, Princeton University Press.
[8] Jones, I. S. F. (1996). "Enhanced carbon-dioxide uptake by the world's oceans." Energy Conversion and Management 37(6-8): 1049 - 1052.
[9] House, K. Z., C. H. House, et al. (2007). "Electrochemical Acceleration of Chemical Weathering as an Energetically Feasible Approach to Mitigating Anthropogenic Climate Change." Environ. Sci. Technol. 41(24).
[10] Drever, J. I., 1988. The Geochemistry of Natural Waters, Prentice Hall, Englewood Cliffs, 437 p.
[11] International Energy Agency 1996a, "Ocean Storage of Co2, Workshop 2, Environmental Impact", IEA Greenhouse Gas Research and Development Programme.
[12] Torvanger A, Kallbekken S. and Rypdal K,:(2004) Prerequisites for Geological Carbon Storage as a Climate Policy Option, Center for International Climate and Environmental Research, Norway.
[13] Dooley, J.J. and Wise, M.A.: 2002, Why injecting CO2 into various geological formation is not the same as climate change mitigation: The issue of leakage, Article presented at Sixth International Conference on Greenhouse Gas Control Technologies, 1-4 October, Kyoto, Japan.
[14] Hawkins, D.G.: 2002, Passing gas: Policy implications of leakage from geologic carbon storage sites, Article presented at Sixth International Conference on Greenhouse Gas Control Technologies, 1-4 October, Kyoto, Japan..
[15] Hepple, R.P. and Benson, S.M.: 2002, Implications of surface seepage on the effectiveness of geological storage of carbon dioxide as a climate mitigation strategy, Article presented at Sixth International Conference on Greenhouse Gas Control Technologies, 1-4 October, Kyoto, Japan
[16] CANA (2004). Carbon Leakage and Geosequestration, Climate Action Network Australia.
[17] Di Fazio does not discuss the affect of population growth on his equations, however it is obvious that WIP and emissions grow for two reasons, per capita industrialisation and growth in the population.
[18] International Energy Agency 1996b, "Technical Responses to Climate Change", IEA Greenhouse Gas Research and Development Program
[19] Seifritz, W. (1990). "CO2 disposal by means of silicates." Nature 345(486).
[20] Dunsmore, H. E. (1992). "A Geological Perspective on Global Warming and The Possibility of Carbon Dioxide Removal as Calcium Carbonate Mineral." Energy Convers. Mgmnt, 33: 565-572.
[21] Lackner, K., C. Wendt, et al. (1995). "Carbon Dioxide Disposal in Carbonate Minerals." Energy 20: 1153 - 1170.
[22] From the Australian Federal department of Industry Science and Tourism in a document titled "Environmental & Economic Life Cycle Costs of Construction, 1998 - Detailed Discussion Paper (section 2 - page 8) and in that document referenced to the Worldwatch Institute.
[23] Prentice, I. C. (2001). Climate Change 2001: The Scientific Basis. Cambridge, UK, Cambridge University Press.
[24] Lovelock, J. E. (1979). Gaia: A New Look at Life on Earth, Oxford University Press.
[25] Vernadsky, V. I. (1924). "La Geochimie." 404pp.
Mitigating The Collapse of Gaia at http://chem11.proboards2.com/index.cgi?board=Discussion&action=print&thread=78
Time Magazine at http://www.time.com/time/specials/2007/article/0,28804,1720049_1720050_1721653,00.html