
Glenorchy
Tasmania 7010 Australia
Phone: 61 3 62497868 (am)
Phone: 61 3 62713000 (pm)
Fax: 61 3 62730010
www.tececo.com
 |
497 Main Road Glenorchy Tasmania 7010 Australia Phone: 61 3 62497868 (am) Phone: 61 3 62713000 (pm) Fax: 61 3 62730010 www.tececo.com |
![]() Printed in cyberspace on recycled electrons
Printed in cyberspace on recycled electrons
Keeping you informed about TecEco sustainability projects. Issue 76, Mar 28, 2008
To all that have written to me in recent months I sincerely apologise if I have not responded. TecEco just do not have the resources to cope and if we spend time replying to email we do not have it for other more important activities.
The economic woes we find ourselves in have taken some of the enthusiasm out of doing anything about global warming. I hear the old rhetoric but see little sense. Global warming will end civilisation as we know it yet we persist in our simplistic analysis of the problem and blunt and ineffective response.
Global response has been underway for some time now and we are tracking on all worst case scenarios. Kyoto policies are just not working which is not at all surprising given the level of understanding of the root cause of the problem, the short term market signals it generates and the constraint mentality amongst politicians. In harder times aspirations are evaporating and constraint going down like a lead balloon particularly with third world countries and the Bush administration in the USA.
The clue the world are missing is plain to see in nature and defined by economics. We have to change the technical basis of our economies in favour of using carbon dioxide as a cost effective input resource. In this way we can harness human nature dominated by selfish desire and get it working for our long term survival.. We can talk about challenge rather than constraint. In this way a real price on carbon would emerge as a result of which people would invent all sorts of way of capturing the gas.
Global warming is a problem of carbon balance not emissions. Nature is a frugal economist and unlike natural economies found in climax eco-systems we are not using as much CO2 as we produce. In the past, before humans began interfering with and now controlling the climate, if the CO2 level was high it would eventually in a self regulatory way be taken up first by plants (on land as well as in the sea) then in soils and sediment and eventually solid carbonate rock. That is why over 4.5 billion years some 40 million gigatonnes of carbonate rocks covering some 8% of the crust has been formed. For life to continue there is a natural progression from life to the earth that upgrades the sequestration permanence of carbon originally from the centre of the earth or that is recycled in the crust.
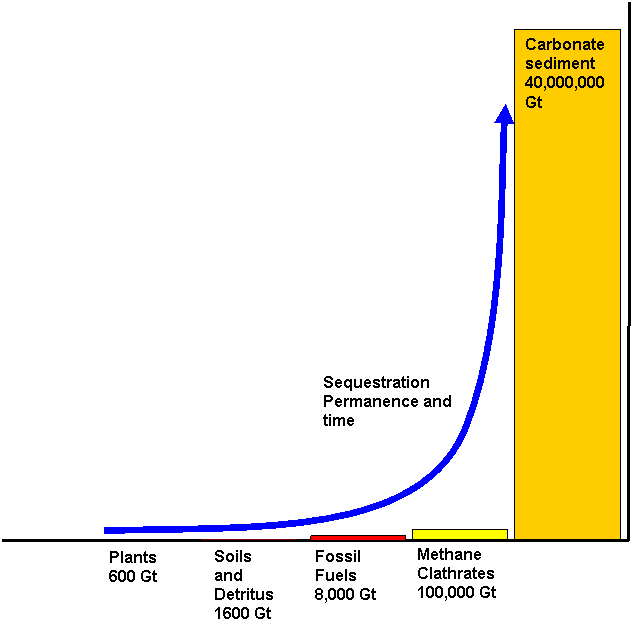
It should be understood from the observation of nature and as a matter of logic that to solve the carbon problem we must accelerate the rate of change in the direction of the blue arrow in the above diagram. Any other strategy such as currently pursued constraint will not hold off inevitable failure given population growth. Constraint is also a negative economic and cannot harness human endeavour the same way as a technical challenge that all can profitably participate in. For long term success the solution to global warming must be profitable and will mainly involve substitution and balance.
To change the balance of carbon in the atmosphere and achieve long term stability we must actually change the materials flowing through the techno-process which is the "material" interface of the economy. That way the underlying molecular flow including the emission of CO2 and other greenhouse gases will change. If 50% of all building and construction were man made carbonate as we advocate the required reduction in atmospheric carbon dioxide would be achieved by 2030 and the problem of global warming solved. Considering that over 70% of materials used in building and construction are mineral based changing 50% to man made carbonate is not impossible to achieve. On the contrary, it is potentially profitable. Once stability is achieved this percentage will have to be monitored and adjusted.
Following this strategy other problems such as that of waste and the supply of fresh water are incidentally solved as a bonus. See Gaia Engineering for details.
The manufacture of salt in dry areas where there is abundant sunshine results in huge deposits of concentrated magnesium chloride as a waste product. This waste product could be used to make magnesium oxide without emissions which in turn could be used to make Eco-Cements.
TecEco are therefore very keen to build a kiln to our proprietary design capable not only of calcination of magnesite but of magnesium chloride salts as found in bitterns.
As some doubt has been expressed that is it possible to convert magnesium chloride to magnesium oxide we draw the attention of readers to several existing processes where this is done. Both are derived from the common Ruthen process whereby hydrochloric acid used for "pickling" steel is recovered with the associated production of iron oxide.
The magnifin process used serpentine as its source of magnesium. The mineral is leached with hydrochloric acid to convert the magnesium silicate to magnesium chloride. This liquor is purified by precipitation and then roasted to produce MgO and HCl which is recycled. If necessary the MgO can then be purified by hydrolysis to produce magnesium hydroxide which can then be calcined to produce magnesium oxide.

Source: FRCA (1998). Flame Retardants 101: Basic Dynamics, CRC Press.
.The Aman process is used by Dead Sea Periclase Ltd. and uses a concentrated MgCl2 brine solution as its magnesium source. The solution is hydropyrolised at around 600 deg. C with steam to produce a mixture of magnesium oxide and hydrochloric acid gas. The oxide produced is treated with water to remove water soluble impurities and the magnesium hydroxide formed is filtered, washed and dried and may be further calcined to produce high quality MgO.
The hydrochloric acid can be recovered and used for other processes such as the Greensols process.

Source: FRCA (1998). Flame Retardants 101: Basic Dynamics, CRC Press.
The Nichromet Process also produces MgCl2 from which MgO and regenerated HCl gas can be produced in a process not dissimilar to the Aman process.
WO0130699 and several other patents describe claimed improved process in which the further benefaction of MgO is not required.
At TecEco we are keen to test our new kiln design with MgCl2. Note that we are also aware that the magnesia we have been supplying is not as well graded as it could be in relation to PC and this results in an unnecessary fineness demand for water in its Tec-Cements. This is yet another reason we are trying to raise money to make our kiln.
If lime and magnesia were made without releases and much less energy then the production and use of major building materials such as cement would be much more sustainable
The Tec-Kiln has been designed for low temperature calcination of alkali metal carbonates and the pyro processing and simultaneous grinding of other minerals such as clays with the above objective in mind.
Hydraulic cements including binders TecEco invented can be made without releases and with much less energy and therefore at lower cost and TecEco are looking for funds to build the first prototype of the kiln.
Roman cements were made from lime and pozzolana (a volcanic ash containing significant quantities of SiO2 and Al2O3) or lime and ground brick and tiles. Learning from them it is possible to split the manufacture of Portland cement into the making of lime and the manufacture of clinker. In this way a lower, more precise amount of non fossil fuel energy can be untilised in a closed system that does not allow releases and much more consistent quality cements produced.
TecEco have demonstrated that reactive magnesia [1] is destined to become a major componenet of hydraulic and hydraulic/carbonating binders of the future and the diagram below depicts the role of the Tec-Kiln for the calcination of magnesite in the thermodynamic cycle of mangesium
Cement made without chemical releases has huge market potential as it represents a solution to the CO2 in the air problem without legacies that is potentially profitable and thus politically acceptable.
In the simplistic representation of the magnesium thermodynamic cycle shown below the TecEco kiln provides de carbonation of MgCO3.
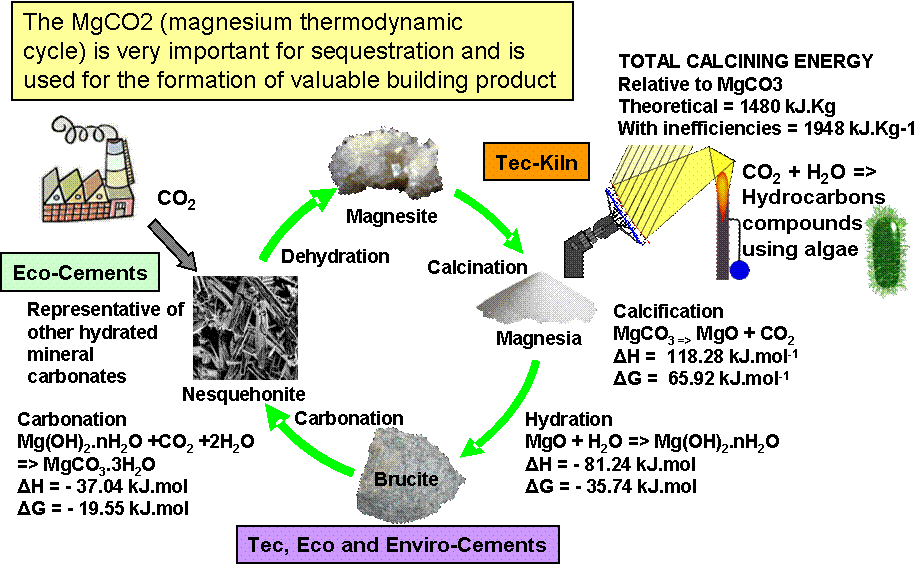
The TecEco Tec-Kiln is an essential part of TecEco's grand plan to sequester massive amounts of CO2 as man made carbonate in the built environment and has the following features: